




In brief, spectroscopy is the study of the electromagnetic spectrum as emitted or reflected by various materials. Reflectance spectroscopy focuses on the radiation reflected by different materials.
Anyone with a little familiarity with rocks can at a glance recognize a piece of basalt or limestone. In this case, color and texture give the observer immediate clues to the rock type. At this simple level a basalt is easily recognized due to its black color (low albedo) and smooth texture (fine-grained). Limestone is buff-white in color (high albedo) and also smooth or fine grained. Sandstones are sometimes red and have noticeable grains. Albedo is simply a measure of the amount of light that is reflected from a surface (rock, mineral, soil, etc). A dark rock only reflects a small portion of sunlight that hits it, thus it has a low albedo. You can think of albedo as the percentage of light that is reflected from a surface versus the amount that shines on the that surface.
However a slate may look very similar to a basalt -- black and fine grained. Sometimes limestone can also look like basalt - when it has small amounts of carbon it too is fine grained and black. Spectroscopy allows scientists to recognize different types of rocks that may initially look or feel the same, or which are too far away to be touched at all, based on subtle differences in how material reflects light across a broad wavelength range.

| 
| 
| 
| 
|
Electromagnetic radiation consists of energy - the shorter the wavelength, the more energetic the light is. Electrons in the rock can absorb this energy, but only at specific energy levels characteristic of a specific atom or molecule. Thus, minerals are more likely to absorb radiation at certain energies, and so only at certain wavelengths. Specific molecules and minerals absorb characteristic wavelengths of radiation, allowing scientists to identify them through their spectra, much as people can be identified through their fingerprints. Energy that is not absorbed is reflected, this reflected energy is what is measured with the spectrometer. We can easily determine the amount of absorption by inspecting dips in the reflectance spectrum.
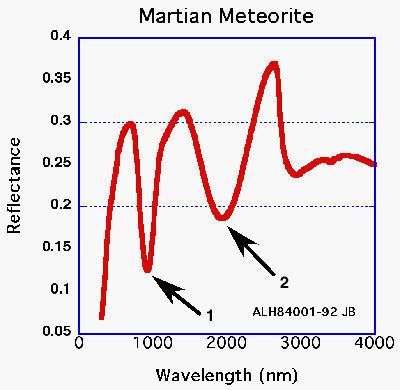 |
The energy reflected by a rock (or from a planetary surface) can be broken into very fine divisions over a wavelength range much wider than that seen by the human eye. This spectrum can be examined using a spectrometer, which can gather data about UV light and IR radiation in addition to the visible ranges (energy, or light, that we can see.) In that spectrum, we should see drops in reflection at certain points where the light is being absorbed by electrons. The points at which these dips, or "absorption bands," occur are characteristic of certain ions, molecules, and minerals. By comparing the position and depth of the absorption bands of an unknown material to the absorption bands of known minerals, we can determine the mineral content of that material. Reflectance spectroscopy allows scientists to estimate the chemical content of planets and asteroids without analyzing actual rock samples.
For the case of basalt as seen by the human eye strong absorptions occur more or less evenly over the whole visible spectrum, thus its black color. In the case of limestone absorptions are weak across the entire visible spectrum, thus its generally light appearance. In the case of basalt atoms of iron occur in several minerals (pyroxenes and olivines) that have a distinctive absorption the near-infrared (~0.950 µm), just beyond the range of the human eye (~0.4-0.7 µm). The exact wavelength center and depth of this absorption indicates the type of pyroxene and the amount of pyroxene relative to olivine (especially with help of another absorption at about 2.0 µm). These two absorptions are often referred to as 1-micron and 2-micron (see arrows 1 and 2 in spectra figure above) features. Unfortunately spectroscopists often mix wavelength units leading to confusion amongst those not familar with this tradition. In the above plot the wavelenghts are shown in nanometers, but here we are referring to microns (1000 nm equals 1 micron)!
The spectral range useful for reflectance spectroscopy for planetary geologists is limited by the wavelengths of sunlight that the planets reflect and by the wavelength of thermal radiation emitted. Planetary bodies eventually emit the energy they have absorbed from light in the form of longer wavelength IR radiation. Reflectance spectroscopy is limited to spectral ranges where the level of reflected light is much greater than the level of emitted light. In total, the useful region for reflectance spectroscopy is usually about .3 to 4.0 µm.
Englert, Peter A. and Pieters, Carle M. (1993) Remote Geochemical Analysis: Elemental and Mineralogical Composition, New York: Cambridge University Press, 312-317.
Rencz, Andrew N.(ed.) (1999) Remote Sensing for the Earth Sciences, New York: John Wiley and Sons, Incorporated.
| CRISM Home Page |
| Spectroscopy of Rocks and Minerals, and Principles of Spectroscopy Roger N. Clark USGS |
| USGS Spectroscopy Page |
| Brown University Reflectance Experiment Lab (RELAB) |